(U-Th)/He thermochronology is based upon the accumulation of 4He in rocks and minerals over geologic time due to the radioactive decay of naturally occurring U, Th, and Sm. Although 4He is constantly being produced by decay, when the rocks are hot the 4He will not be retained in the crystals, and will instead diffuse out of the system (and into the atmosphere, where it makes up about 1 ppm, or 0.0001%, of what we breathe).
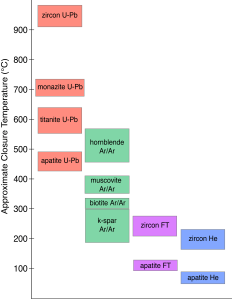
However, once a mineral is sufficiently cool, the 4He will be retained. The transitional temperature is called the closure temperature, and is different depending upon a variety of factors, including what mineral you are interested in. For example, 4He is typically only retained in apatite when the crystal cools below about 60–70°C, or in undamaged zircon when it cools below ~185°C. Because the earth gets hotter as you go deeper, the cooling history of a rock often tracks its path from the deep earth up to the surface.
Thermochronologists exploit these phenomena to constrain a wide range of geologic problems, including the formation and evolution of mountain belts, deep-time landscape evolution , and the temporal and spatial evolution of fault systems.
Since the late 1990’s, (U-Th)/He dating has become a popular and widely applicable technique capable of helping answer a wide range of geologic questions. A short list of recent applications includes:
- Uplift and Exhumation over both short and long timescales
- Landscape evolution
- Eruption and Mineralization ages
- Detrital Thermochronology
- Reheating and Basin Thermal Histories
- Impact chronology
- Meteorite thermochronology
- Wildfire chronology
- Dating of thermal resetting events like basalt flows and pseudotachylite formation
If you would like to read more about the (U-Th)/He technique, or about thermochronology in general, please link through to the world’s foremost thermochronology blog. Check out this entry describing how to pick and select apatites for (U-Th)/He Thermochronology!
At CU we are equipped to measure (U-Th)/He and U-Pb ages on a variety of materials and would love to help you with your thermo- and geo-chronology needs. We are also always interested in developing new collaborative relationships. Please feel free to contact either Professor Becky Flowers (lab PI) or Jim Metcalf (lab manager) with questions.
**In 2022 Becky spearheaded an effort to publish two papers that provide an in-depth overview of the (U-Th)/He method. These are both provided free of charge from GSA Bulletin, and features input from both Jim and a whole pile of actually well-known co-authors. You can find part 1 here and part 2 here!

